Obstructing the Right to Know
CHEMICAL SELECTION
The largest study of SDS accuracy in the peer reviewed literature was done in 1995 and analyzed 150 SDSs. In this pilot project, the BlueGreen Alliance and Clearya set out to automate the review process to produce a larger initial data set. We also wanted to determine what would be needed to fully automate searches for SDS accuracy.
To select chemicals for this investigation, we identified chemicals in the Healthy Building Network (HBN) Pharos database that have been designated by authoritative bodies as carcinogens, mutagens, or reproductive toxicants. We selected a subset of
100 chemicals for which at least three authoritative entities designated the same hazards. The authoritative bodies lists include:
- The European Council Regulations No 1272/2008 (REACH);
- The Hazardous Chemical Information System (HCIS) from Safe Work Australia;
- The S. National Library of Medicine’s (NLM) Hazardous Substances Data Bank;
- The Chemical Management Center of Japan’s National Institute of Technology and Evaluation (NITE-CMC);
- The U.S. National Toxicology Program’s Report on Carcinogens (NTP ROC);
- The NTP Reports on Developmental and Reproductive Toxicity; and
- The California Office of Environmental Health Hazard Assessment (Prop 65).
Ten of the chemical substances included in our project are among the first 33 High Priority Substances being reviewed by the EPA under TSCA. We specifically included well-studied substances such as benzene and formaldehyde to determine if inaccuracies also exist in SDSs for chemicals with extensive evidence of hazard.
We excluded chemicals based on the following criteria:
- Low Chemical Domestic Production/Import Volume. A chemical was determined to be less relevant for our study if production volume was relatively low (i.e. < 1 million pounds of production/import) based on EPA’s consumer data reporting (CDR).
- Low number of English We excluded chemicals with few English SDSs present in the ChemWatch database (i.e. fewer than 30 active SDSs for this specific substance). Thirty SDSs per chemical were chosen for our exclusion criteria to preliminarily ensure there were a reasonable number of non-duplicative SDS options to choose from given our 1000 SDS limit.
- Irrelevant in Practice. We excluded some chemicals because they are not widely used. This was based on industry knowledge from several subject matter experts in environmental and occupational health and science policy.
We aimed to have a slightly higher number of major chemical manufacturers’ SDSs than SDSs from research and development (R&D) labs for a few specific reasons: first, to ensure good representation of SDSs in current use in U.S. workplaces; second to understand how SDS inaccuracies compare between R&D labs and large chemical producers; and third to identify industry specific trends worth exploring in the next phase of this project.
The screening process resulted in an analysis of 34 chemicals in 655 SDSs. The concentration of the 34 evaluated chemicals in the 655 SDSs met the cut-off limit specified by the U.S. OSHA and the GHS for which hazard reporting is required. To validate the relevance of the selection process, we reviewed the International Chemical Safety Cards (ICSCs) produced by the WHO and the ILO, with the cooperation of the European Commission. There are ICSCs for 1700 chemicals out of the 45,000 in commerce. Twenty nine of the 34 chemicals in this pilot study have ICSCs.
ANALYSIS
Clearya developed software to automate the process of screening tens of thousands of SDSs for accuracy. The software can identify chemical hazard classifications missing from SDSs for chemicals that may cause cancer, birth defects, and other harm. Additionally, it can detect instances of misrepresented health hazards, such as a SDS that describes a known hazard as merely suspected. SDS content was extracted from PDF formats using ChemParser.

This SDS is provided for methylmercury chloride. There is no controversy that this substance is suspected of causing cancer, can interfere with normal fetal development, and is suspected of damaging fertility. Yet, these warnings, as well as their associated pictograms, are missing from the Hazards Identification section and from the Composition/Information on Ingredients sections, which should list all hazard classifications for substances included in this product.
FINDINGS
Our analysis of an initial set of 655 SDSs found that errors in SDSs are common: 30% of the SDSs included inaccurate chemical hazard warnings.
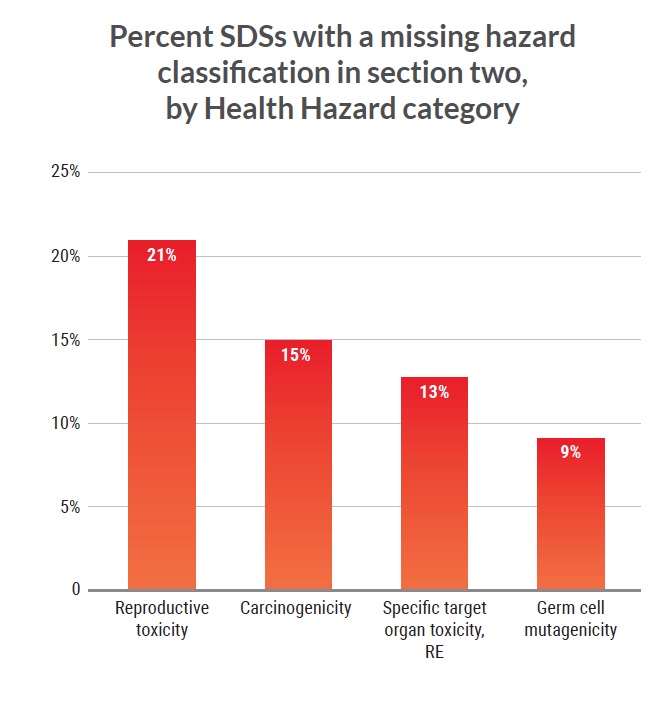
Findings by health hazard
For the 30 prioritized carcinogenic (cancer-causing) substances present in 512 of the SDSs we obtained, 15% of SDSs failed to report carcinogenicity in the Hazards Identification section (Section 2). Of the 172 SDSs with mutagenic substances (i.e., substances that may cause genetic mutations), 9% failed to report this hazard. The findings were as concerning in the SDSs for substances that are toxic to human reproduction (i.e., harming fertility or fetal development), and for
substances with specific target organ toxicity. We found 21% of 372 SDSs with chemicals toxic to reproduction lacked warnings for this hazard and 13% of 278 SDSs with chemicals of specific organ target toxicity either omitted or showed inaccurate warnings for this hazard.
One telling example of these errors is found in an SDS for vinyl chloride, a known human carcinogen. The SDS defined vinyl chloride as a chemical that causes skin, eye, and respiratory irritation; it lacked any mention
of carcinogenicity. Another SDS for benzene, which should warn of its mutagenicity, carcinogenicity, and specific target organ toxicity, only reported skin and eye irritation and harmfulness if swallowed, contacted with skin, or inhaled. It failed to mention the other highly hazardous effects of this well-studied chemical.
Findings by substance
Looking at the 34 prioritized substances for which SDSs were obtained, the findings are disturbing: 88% of the substances had at least one SDS file with missing hazards in the Hazards Identification section (Section 2 of the SDS). Two examples are 1,3-butadiene—for which 75% of SDSs (15/20) failed to report reproductive toxicity—and diethylhexyl phthalate (DEHP), for which 57% of SDSs (16/28) failed to report carcinogenicity.
Findings by chemical manufacturer
The SDSs in our sample were produced by 16 of the largest international chemical firms as well as small and medium sized producers. The results show that 34% (97/289) of the chemical producers supplied SDS files with inaccurate hazard statements. Of the 33 chemical producers with at least five SDSs, the worst performing one supplied 100% (6/6) inaccurate SDSs while the average chemical producer supplied 26% inaccurate SDSs.
Findings for selected large chemical producers
To examine whether reporting inaccuracies characterize only smaller SDS producers, we repeated the analysis for a sample of 34 SDSs by three large producers: 3M, BASF, and Covestro.
Similar patterns were found in that analysis:
- We found that SDSs failed to report accurate hazards in 11% of SDSs for substances of specific target organ toxicity (2/18) and 23% of SDSs with substances toxic to reproduction (7/30).
- Among the prioritized substances included in these SDSs, 33% (3/9) had at least one SDS file with missing hazards in the Hazards Identification section (Section 2).
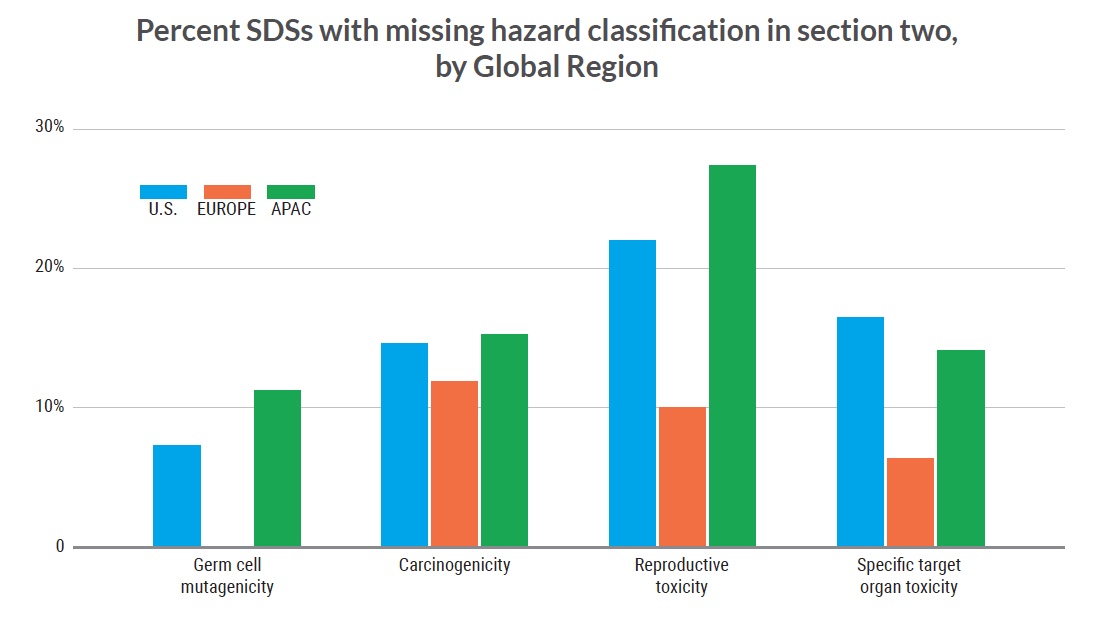
Findings for different regions
To examine whether the patterns found in our analysis are similar across SDSs from companies in different regions , the analysis was repeated separately for SDSs in the following regions: Europe (147 SDSs), the
U.S. (228 SDSs), and Asia-Pacific (236 SDSs).
Our findings show accuracy rates for the four hazard categories vary by region:
- Carcinogenicity was missing in the Hazards Identification section in 15% of SDSs from the S.; 15% of Asia-Pacific SDSs; and 12% of Europe SDSs.
- Reproductive toxicity hazard was missing from 22% of S. SDSs; 27% of Asia-Pacific SDSs; and 10%of Europe SDSs.
- Specific target organ toxicity hazard was missing from 16% of S. SDSs; 14% of Asia-Pacific SDSs; and 6% of Europe SDSs.
- Mutagenicity hazard was missing from 7% of S. SDSs and 11% of Asia-Pacific SDSs.